S-Monovette® EDTA K2E, 2.7 ml, cap violet, (LxØ): 75 x 13 mm, with paper label
- Specifications
- Brochures
- FAQ
- Videos
- Manuals
- Literature
- Instructions for Use
- Declarations of conformity
Product description
| Order number | 04.1915.100 |
|---|---|
| Product description | S-Monovette® EDTA K2E, preparation: K2 EDTA, 2.7 ml, membrane screw cap, cap violet, colour code ISO, (LxØ) without cap: 75 x 13 mm, with paper label, label/print: violet, 50 piece(s)/case, sterile |
Product characteristics
| Type of collection | venous |
|---|---|
| Type of preparation | K2 EDTA |
| Preparation concentration | 1.6 mg/ml blood |
| Colour code | ISO |
| Label/ Print | with paper label |
| Colour of print/label | violet |
| Graduation | fill mark |
| Cap | membrane screw cap |
| Closure type | screw cap |
| Liquid-tight | yes |
| Connection | membrane screw cap |
Size
| Sample volume | 2.7 ml |
|---|---|
| Diameter | 13 mm |
| Length including cap | 91 mm |
| Length excluding cap | 75 mm |
| Length including cap and plunger | 112 mm |
Material & colours
| Colour of product | transparent |
|---|---|
| Closure material | High Density Polyethylene (HD-PE) |
| Colour of cap | violet |
| Membrane material | rubber |
| Tube material | Polypropylene (PP) |
| Piston material | High Density Polyethylene (HD-PE) |
| Piston plunger material | Polystyrene (PS) |
Purity & certification
| Satisfies the requirement | IATA, ADR |
|---|---|
| Product category | In vitrodiagnostic device, CE |
| CE certified | CE |
| Purity standard | sterile |
| Sterilisation | Electron irradiation |
| Batched | yes |
Packaging
| Minimum order qty. | 500 |
|---|---|
| Type of smallest subpackaging | case |
| Piece(s) / inner box | 50 |
| Piece (s) / outer case | 500 |
| Piece(s) / pallet | 39000 |
| Depth of box | 171 mm |
| Width of box | 116 mm |
| Height of box | 73 mm |
| Depth of case | 388 mm |
| Width of case | 358 mm |
| Height of case | 135 mm |
| Case volume | 0.0188 cbm |
| Weight of product | 0.0042 kg |
| Weight of case | 2.86 kg |
| EAN of inner box | 4038917382250 |
| EAN case | 4038917382243 |


Gentle to animals - Ordering information for pet blood collection products
Gentle to animals - Ordering information for pet blood collection products
Language:
Go for Gold! Short
Go for Gold! Short
Language:
Basics of venous blood collection
Basics of venous blood collection
Language:

Go for Gold!
Go for Gold!
Language:

Gentle to animals
Gentle to animals
Language:

S-Monovette® - The sustainable blood collection system
S-Monovette® - The sustainable blood collection system
Language:

Preanalytical Workflow
Preanalytical Workflow
Language:

S-Monovette® Serum Gel LightPROTECT
S-Monovette® Serum Gel LightPROTECT
Language:

Synergie S-Monovette® & Tempus600®
Synergie S-Monovette® & Tempus600®
Language:
S-Monovette® cfDNA Exact
S-Monovette® cfDNA Exact
Language:

S-Monovette® RNA Exact - Freezing conditions
S-Monovette® RNA Exact - Freezing conditions
Language:

S-Monovette® RNA Exact
S-Monovette® RNA Exact
Language:

S-Monovette Citrate Tube

S-Monovette® Lithium-Heparin Gel+
S-Monovette® Lithium-Heparin Gel+
Language:

Colour coding in blood collection
Colour coding in blood collection
Language:
Centrifugation
Centrifugation
Language:

S-Monovette® Hirudin 1.6 ml

Blood collection systems for paediatrics
Blood collection systems for paediatrics
Language:

PBM PatientBloodManagement
PBM PatientBloodManagement
Language:

Tips & Techniques in Preanalytics
Tips & Techniques in Preanalytics
Language:
S-Monovette® DNA Exact
S-Monovette® DNA Exact
Language:

Single-Use Tourniquet
Single-Use Tourniquet
Language:

Veterinary Medicine
Veterinary Medicine
Language:
Micro-Needle for Veterinary Medicine
Micro-Needle for Veterinary Medicine
Language:

Blood Collection Systems
Blood Collection Systems
Language:

Ordering Information Blood Collection Systems
Ordering Information Blood Collection Systems
Language:

Safety Products
Safety Products
Language:
S-Monovette® GlucoEXACT Laboratory
S-Monovette® GlucoEXACT Laboratory
Language:

S-Monovette® HCY-Z-Gel
S-Monovette® HCY-Z-Gel
Language:
S-Monovette® ThromboExact
EDTA-Anticoagulants
General
-
+
Which sedimentation pipette is suitable for the S-Sedivette®?
For ESR determination using the S-Sedivette® a sedimentation pipette is not required. Blood collection, transport and measurement can be safely carried out in the enclosed blood collection tube, the S-Sedivette®.
-
+
Why does the S-Monovette® have a screw cap?
The screw cap enables a gentle opening of the tubes and minimises the aerosol effect which occurs when using stoppers.
-
+
Is there an S-Monovette® without additives?
Yes, the neutral S-Monovette® is free from additives and is available in various different designs.
-
+
Does the S-Monovette® meet the IATA guidelines on shipping samples using air freight?
Yes, the S-Monovette® meets the requirements of the IATA guidelines and is impervious to fluids at a pressure difference of 0.95 kPa.
-
+
What storage conditions are recommended for S-Monovettes before blood collection?
We recommend storage at room temperature.
-
+
What material is the S-Monovette® made from?
The S-Monovette® is made entirely from plastic. It is therefore particularly unbreakable and ideally suited for transport. The main components are the tube made from polypropylene (PP), the membrane screw cap made from polyethylene (PE) and natural rubber and the plungers made from polyethylene (PE).
-
+
How are the S-Monovettes packed?
The S-Monovette® is packed in a cardboard box filled with 50 items as standard. This enables low levels of waste generation and simple disposal.
-
+
What material is the membranes of the screw cap made from and why is it lower down?
The membrane is made from natural rubber and is embedded lower down in the screw cap in order to prevent direct contact with the puncture site. The result is improved infection protection, allowing the user to work safely with the S-Monovette®.
-
+
Does it have an expiry date and, if yes, where can it be found?
An expiry date is necessary in order to rule out any influence caused by the ageing process and in order to ensure a consistently high sample quality. This is indicated as the month and year and can be found on both the product and the packaging.
-
+
How is the S-Monovette® certified?
The S-Monovette® is classified as an in vitro diagnostic tool and is CE marked. The entire quality management system is certified in accordance with EN ISO 13485.
-
+
Does the age of the S-Monovette® have an impact on the nominal volume?
The age of the S-Monovette® has no impact on the filling volume. The S-Monovette® is designed so that 100% of the nominal volume can always be obtained.
-
+
Is there a blood collection tube with a particularly low volume for venous blood collection?
Yes, the paediatric S-Monovette® is available in volumes 1.1 ml–1.4 ml, depending on the preparation.
Pre-analytics
-
+
What influencing factors are there?
The patient should sit for at least 10 minutes before blood collection, stasis time, puncture, collection order, correct filling (citrate, EDTA), mixing the sample, storage and transport conditions (e.g. temperature), blood collection: selection of method – aspiration or vacuum.
-
+
Why is haemolysis particularly significant?
Depending on the degree of haemolysis, haemolytic samples distort or prevent the determination of analytes and the blood has to be collected again. Here are a few example of parameters which can be affected: LDH, K, AST (GOT), ALT (GPT), troponin, ß-HCG, glucose, CK, PT, aPTT, D-dimer.
-
+
How can I prevent haemolysis?
Here are some of the most important influencing factors for preventing haemolytic samples: avoid vigorous shaking, prevent heavy vibrations during transport of the blood sample, when collecting blood in particular from inserted catheters only collect blood by means of aspiration, use needles with large lumens.
-
+
Can the platelets be activated during blood collection? If so, how can this effect be reduced?
A slower, more gentle blood flow can be achieved by choosing aspiration as a blood collection technique. This also causes the smallest possible effect on the platelets.
-
+
In centrifugation, is there a difference between the relative centrifugal force and the number of rotations?
Relative centrifugal force (RCF) is the ratio between the number of rotations and the radius of the centrifuge. The values can be calculated from one another using the formula: RCF = 1.118*10-6*r*n2. r = radius of the centrifuge, n = number of rotations (= rotations/min), g = 9.81 m/s2 (= gravity), RCF = relative centrifugal force.
-
+
What is the difference between a centrifuge with a fixed-angle rotor and a centrifuge with a swing-out rotor?
The inserts in the fixed-angle rotor are at a fixed angle to the axis of rotation (often 30°) and remain at this angle during centrifugation. In contrast to this, the inserts in the swing-out rotor move into a position perpendicular to the axis of rotation at the start of centrifugation and remain there. This is the only way to achieve an optimal gel separating layer.
-
+
What can I do if the vein conditions are poor?
Carry out blood collection using the aspiration technique. An optimal solution is to collect blood using a Safety-Multifly® needle and S-Monovette®.
-
+
Does the position of the patient matter?
Yes, very much so. Blood should be collected from the patient when he or she is in a sitting or lying position. Before blood collection, the patient should rest sit for 10 minutes and avoid physical activity.
-
+
What role does the time of blood collection play?
The time of blood collection plays an important role for many laboratory parameters, so blood should normally be collected in the morning between 7 a.m. and 9 a.m. from patients with an empty stomach (drinking water is permitted).
-
+
Where can I find more information on pre-analytics?
You can find more information on our website in the Service section, under Publications.
-
+
How does haemolysis occur?
Haemolysis occurs as a result of damage to the cell membrane of erythrocytes. The red haemoglobin (main component of red blood cells) which is then released can disrupt the analysis of various different parameters, depending on the amount released.
-
+
Why do citrate S-Monovettes have to be filled up to the fill mark?
Underfilling can cause an incorrect ratio between the citrate presented and the blood and can therefore lead to incorrect results.
Glucose
-
+
Does the S-Monovette® Glucose have to be mixed?
The S-Monovette® Glucose must be carefully inverted in order to ensure optimal distribution of the preparation in the blood.
-
+
What is the shelf life of the S-Monovette® Glucose?
The shelf life of the S-Monovette® Glucose is 12 months.
-
+
How is the S-Monovette® Glucose prepared?
The S-Monovette® Glucose is prepared with fluoride and EDTA.
-
+
How is the S-Monovette® GlucoEXACT prepared?
The S-Monovette® GlucoEXACT is prepared with citrate and fluoride.
-
+
What is the difference between the S-Monovette® Glucose and the S-Monovette® GlucoEXACT?
The two S-Monovettes differ in terms of their preparation and, as a result of this, in terms of the start of glycolysis inhibition. Glycolysis inhibition occurs in the S-Monovette® GlucoEXACT, which is filled with blood, immediately after the blood is collected. In contrast, the inhibition in the S-Monovette® which is only prepared with fluoride starts after approximately two hours.
Serum/plasma
-
+
When should a gel preparation be used?
A gel preparation should be used when a stable barrier between the blood clot and the serum and between the blood cells and the plasma is needed for transport and storage. The gel's thickness means that during centrifugation it forms a separating layer between the blood clot and the serum or the blood cells and the plasma, providing a stable barrier for transport and strorage.
-
+
How quickly can the S-Monovette® Serum/Serum Gel be centrifuged after the blood is collected?
For optimal serum quality, we recommend that the S-Monovette® Serum/Serum Gel be stored standing upright for at least 30 minutes before being centrifuged.
-
+
Can an S-Monovette® which is prepared with gel be re-centrifuged?
Once the gel barrier has formed, further centrifugation is not recommended as the barrier effect of the gel separating layer would no longer be guaranteed.
-
+
Is there a clotting activator in the S-Monovette® Serum?
Yes, there is a silicate on the granulate beads in the S-Monovettes.
-
+
What material is the gel made from?
The gel in the S-Monovette® is made from a polyacrylic ester which, due to its thickness, forms a stable separating layer during centrifugation.
-
+
What should be taken into account when centrifuging S-Monovettes with gel?
In order to achieve an evenly formed separating layer, centrifugation must be carried out in a centrifuge with a swing-out rotor. The S-Monovette® Serum-Gel is to be centrifuged at 2500 xg for 10 minutes and the S-Monovette® Plasma-Gel at 2500 xg for 15 minutes or 3000 xg for 10 minutes, both at room temperature.
-
+
Why do the S-Monovette® Serum and the S-Monovette® Plasma contain plastic granulate?
The plastic granulate is coated with the respective preparation and supports the mixing process between the blood and the preparation.
-
+
Can heavy metals be determined using the S-Monovette® Serum or S-Monovette® Plasma?
The S-Monovette® Metal Analysis (product number 01.1604.400) in combination with the Safety-S-Monovette® Needle for Metal Analysis (product number 85.1162.600) is recommended for the determination of heavy metals. The S-Monovette® Serum and S-Monovette® Heparin have not been evaluated for the determination of heavy metals.
-
+
What is the shelf life of the S-Monovette® Serum?
The shelf life of the S-Monovette® Serum is 36 months.
-
+
What is the shelf life of the S-Monovette® Lithium-Heparin?
The shelf life of the S-Monovette® Lithium-Heparin is 36 months.
Citrate
-
+
What is the minimum quantity of blood which must be filled in the S-Monovette® Citrate?
An exact filling volume and therefore a correct mixing ratio is recommended to avoid incorrect measurements due to underfilling.
-
+
How is the S-Monovette® Citrate prepared?
The S-Monovette® Citrate is prepared with trisodium citrate solution which makes up 10% of the nominal volume. This achieves the ratio which is important for clotting diagnostics – 1 part citrate to 9 parts blood.
-
+
What is the recommended centrifugation for the S-Monovette® Citrate?
The recommended centrifugation for the S-Monovette® Citrate is 1800 x g for 10 minutes at room temperature.
-
+
Does the S-Monovette® Citrate have to be mixed after the blood has been collected?
The S-Monovette® Citrate should be carefully inverted 8–10 times after the blood has been collected.
-
+
What needs to be taken into account when collecting blood using the Safety-Multifly® needle?
When taking blood using a Safety-Multifly® needle, blood remains in the tube (known as dead volume), reducing the volume of blood in the first S-Monovette®. If only one S-Monovette® Citrate is to be collected at first or in total, carrying out the preceding collection using an empty tube is recommended to avoid underfilling.
-
+
What is the shelf life of the S-Monovette® Citrate?
The shelf life of the S-Monovette® Citrate is 15 months.
EDTA
-
+
Can virus diagnostics be measured from EDTA Gel S-Monovettes?
Studies have shown a significantly improved recovery rate in EDTA Gel S-Monovettes compared to EDTA S-Monovettes.
-
+
What does EDTA K3 stand for?
EDTA K3 stands for ethylenediaminetetraacetate tripotassium. It binds calcium and prevents clotting. The blood sample remains liquid.
-
+
How is the S-Monovette® EDTA prepared?
The S-Monovette® EDTA is prepared with K3 EDTA.
-
+
What is the shelf life of the S-Monovette® EDTA?
The shelf life of the S-Monovette® EDTA is 18 months.
-
+
What is recommended for patients with anticoagulant-induced pseudothrombocytopenia?
The S-Monovette® ThromboExact is recommended for these patients.
-
+
Does the S-Monovette® EDTA have to be mixed?
The S-Monovette® EDTA must be carefully inverted in order to ensure optimal distribution of the preparation in the blood.
Special
-
+
Which S-Monovette® is recommended for oGTTs?
The S-Monovette® GlucoEXACT is recommended.
-
+
Which S-Monovette® can be used to determine heavy metals?
The S-Monovette® Metal Analysis (product number 01.1604.400) in combination with the Safety-Needle for Metal Analysis (product number 85.1162.600) is recommended for the determination of heavy metals. The normal S-Monovette® Serum and S-Monovette® Heparin have not been evaluated for the determination of heavy metals.
-
+
Can an anticoagulant (EDTA, citrate or heparin)-induced pseudothrombocytopenia be determined using the S-Monovette® ThromboExact?
Yes, the S-Monovette® ThromboExact has been specially developed for anticoagulant-induced pseudothrombocytopenia. If the thrombocyte count is low in EDTA blood, the S-Monovette® ThromboExact can be used to find out whether the thrombocyte count is a false low result and the patient is experiencing anticoagulant-induced pseudothrombocytopenia or whether the thrombocyte count really is low.
-
+
Can the S-Monovette® CTAD be used for routine clotting diagnostics?
The following routine clotting parameters can be measured using the S-Monovette® CTAD: aPTT, PTT (Quick), fibrinogen, thrombin time, AT3 and Protein C. Individual tests must be carried out for all further parameters.
-
+
Which S-Monovette® is recommended for thrombocyte function diagnostics?
The S-Monovette® Hirudin which was developed for the Multiplate device by Roche (formerly Verum Diagnostica) and the S-Monovette® PFA100 which was designed for the PFA100/PFA200 device by Siemens are recommended here. Both S-Monovettes can be used for the special thrombocyte function diagnostics.
-
+
Which S-Monovette® is recommended for the determination of homocysteine?
The S-Monovette® Homocysteine HCY-Z-Gel is recommended, which stabilises the HCY concentration up to 96 hours after the blood is collected.
We would like to show you additional content which is hosted by a third party. For this purpose we need your consent to set cookies:

User Guide S-Monovette® EDTA
User Guide S-Monovette® EDTA
Language:

S-Monovette® - New marking of the product labels (EU-code)
S-Monovette® - New marking of the product labels (EU-code)
Language:

S-Monovette® - New marking of the product labels (ISO-code)
S-Monovette® - New marking of the product labels (ISO-code)
Language:
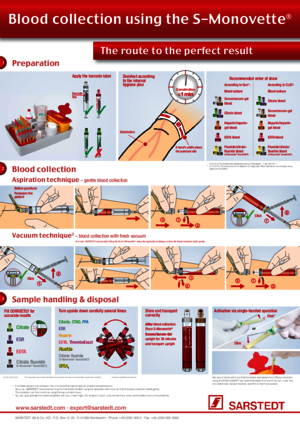
Blood Collection with the S-Monovette®
Blood Collection with the S-Monovette®
Language:

Einfrierbedingungen S-Monovette® RNA Exact
Einfrierbedingungen S-Monovette® RNA Exact
Language:

S-Monovette® Freezing Conditions
S-Monovette® Freezing Conditions
Language:

Handhabung S-Monovette® Citrat

S-Monovette® - Blutentnahmereihenfolge

S-Monovette®
S-Monovette®
Language:
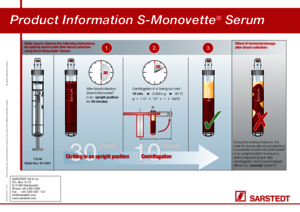
Product Information S-Monovette® Serum
Product Information S-Monovette® Serum
Language:

S-Monovette®
S-Monovette®
Language:
S-Monovette®
S-Monovette®
Language:

User Guide Cool and warm transport container
User Guide Cool and warm transport container
Language:

Correct application of barcode label to S-Monovette®
Correct application of barcode label to S-Monovette®
Language:

Simultaneous extraction of mRNA and microRNA with the Norgen Total RNA Purification Kit Dx (REF Dx17200) from S-Monovette® RNA Exact
Simultaneous extraction of mRNA and microRNA with the Norgen Total RNA Purification Kit Dx (REF Dx17200) from S-Monovette® RNA Exact
Language:

Simultaneous extraction of mRNA and microRNA with the Maxwell® CSC RNA Blood Kit from S-Monovette® RNA Exact
Simultaneous extraction of mRNA and microRNA with the Maxwell® CSC RNA Blood Kit from S-Monovette® RNA Exact
Language:

Simultaneous extraction of mRNA and microRNA with the MagMAX™ mirVana™ Total RNA Isolation Kit from S-Monovette® RNA Exact
Simultaneous extraction of mRNA and microRNA with the MagMAX™ mirVana™ Total RNA Isolation Kit from S-Monovette® RNA Exact
Language:

Application Note MACHEREY-NAGEL NucleoSpin Blood Kit

Application Note MagMax™ Cell-Free DNA Isolation Kit

Application Note Zymo – Quick-cfDNA Serum & Plasma Kit

Application Note MACHEREY-NAGEL – NucleoSnap® cfDNA Plasma-Kit
Application Note MACHEREY-NAGEL – NucleoSnap® cfDNA Plasma-Kit
Language:

Application Note Promega GmbH – Maxwell® RSC ccfDNA LV Plasma Kit
Application Note Promega GmbH – Maxwell® RSC ccfDNA LV Plasma Kit
Language:

Application Note MACHEREY-NAGEL – NucleoMag® cfDNA kit

Sarstedt S-Monovette® CPDA

Comparison of S-Monovette® cfDNA Exact with two blood collection tubes for stabilization of cfDNA
Comparison of S-Monovette® cfDNA Exact with two blood collection tubes for stabilization of cfDNA
Language:

Whitepaper S-Monovette® LH Gel+ ROCHE cobas systems V1.0

Application Note S-Monovette® RNA Exact

Whitepaper S-Monovette® LH Gel+ Siemens Atellica V1.0
Application Note chemagic™ Total RNA 9k Kit H24

Whitepaper S-Monovette® RNA Exact

Technical Bulletin S-Monovette® RNA Exact

Application Note NucleoSpin® RNA Blood Midi

Application Note InviMag® Blood RNA Exact Kit/ IG & S-Monovette® RNA Exact
Application Note InviMag® Blood RNA Exact Kit/ IG & S-Monovette® RNA Exact
Language:

Whitepaper S-Monovette® GlucoEXACT 2016

Whitepaper S-Monovette® DNA Exact

Comparison of S-Monovette® cfDNA Exact with two blood collection tubes for stabilization of cfDNA
Comparison of S-Monovette® cfDNA Exact with two blood collection tubes for stabilization of cfDNA
Language:

SARSTEDT S-Monovette® ESR Blood Collection System

SARSTEDT Monovette® VD and VD screw top cup

SARSTEDT Adapter cap for S-Monovette®

SARSTEDT Blood Gas Monovette® and accessories

S-Monovette® cfDNA Exact

S-Monovette® RNA Exact

SARSTEDT S-Monovette® Blood collection system

S-Monovette®, Monovette®, S-Sedivette® (sterile)
S-Monovette®, Monovette®, S-Sedivette® (sterile)
Downloads: